
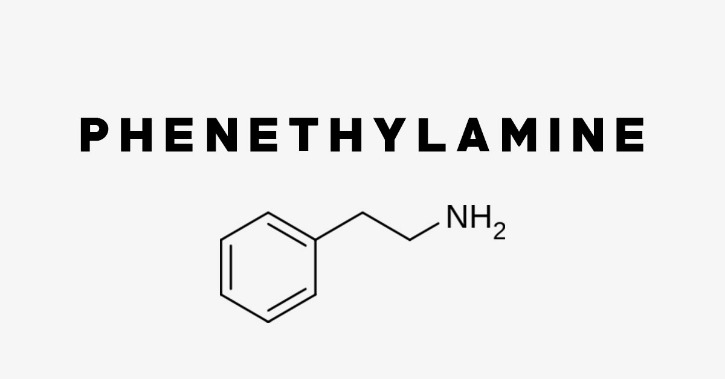
Phenethylamine is a chemical substance that naturally exists in the body. Phenethylamine is produced by various species in the human, plant, and animal kingdoms. Phenethylamine is used for depression, weight loss, sports performance, and to improve mood and concentration. Psychedelic phenethylamines commonly act to increase neurotransmitter levels, oftentimes by interrupting storage of neurotransmitters in vesicles and reversing the flow the neurotransmitter reuptake pumps. Some may appreciably block neurotransmitter reuptake themselves or have inhibitory activity at monoamine oxidase. Phenylpropanolamines are β-hydroxy (β-OH) analogs of phenylisopropylamines.
Figure 5

But unlike stimulant drugs like amphetamine, which release a flood of these neurotransmitters in an uncontrolled manner. Food training and catheter implantation surgery were performed as described previously 48. The timeline for self-administration procedures is illustrated in Figure 4A.
Recent Activity

It was findings, such as these that were partly responsible for our continued interest in this area. To further investigate this unique and very intriguing phenomenon, we trained rats to discriminate racemic MDA from saline vehicle. This seemed to be borne out by subsequent stimulus generalization studies. The phenyethylamine moiety has risen from humble beginnings as a presumed metabolic by-product to serve as a noteworthy scaffold for therapeutically useful agents, and for an even greater number of abused substances.

The actions on the monoamine systems (serotonin, norepinephrine, dopamine) differ between substances, ultimately causing variations in subjective experience, safety, and addictive potential. Psychedelic phenethylamines often comes in a ‘series’ of compounds (e.g. DOx, 2Cx, 25x-NBOMe). Psychedelics phenethylamines have a wide variety of target receptors, although commonly have serotonergic action and it could be considered a pharmacologic definition of their class that they have activity or bind with 5HT2A receptors.
The solubilized protein samples were stored in a deep freezer before use. One study found that supplementing with 10–60 milligrams of phenethylamine daily along with the antidepressant drug called selegiline (Anipryl, Eldepryl) helped relieve depression in 60 percent of participants. An impressive 86 percent experienced relief from depression symptoms for up to 50 weeks.
Take Your Time:
- USVs recording is a useful method to measure changes in emotional state caused by exposure to drugs such as cocaine or methamphetamine 49,50.
- In addition to being naturally created by the human body from the amino acid phenylalanine, it can also be found in trace levels in some fermented foods, such as chocolate, cheese, and other foods.
- These products should be avoided until more safety data become available.
- Many of these psychoactive compounds exert their pharmacological effects primarily by modulating monoamine neurotransmitter systems; however, there is no mechanism of action or biological target that is common to all members of this subclass.
- Dopamine is simply phenethylamine with a hydroxyl group attached to the 3 and 4 position of the benzene ring.
- In particular, collaborators Dr. Richard Young and Dr. Małgorzata Dukat deserve special recognition for their critical scientific input and assistance with many of these studies.
The appropriate dosage of phenethylamine for nootropic use can vary based on individual needs, tolerance, and goals. Standard dosages for cognitive enhancement and mood support generally range between 100 mg and 500 mg per day. It is important to start with a lower dose to assess tolerance, especially since PEA’s effects can be potent and short-lived. In the body, phenethylamine is rapidly broken down by the enzyme monoamine oxidase B (MAO-B).
David Tomen
Note that the numbers in parentheses (1, 2, etc.) are clickable links to these studies. Three deaths have been linked to 2C-T-7, one death to 2C-T-21, two deaths to 2C-I-NBOMe, and one death to 2C-E 34–41. It should be noted that there are recent reports of poor clinical outcomes after exposure to unconfirmed 2Cs, including presumed 2C-I-NBOMe (25i). Other published case reports of 2C intoxication are listed in Table 3 41–43.
Upgrade Your Brain
One reason might be that they are unable to readily cross the blood–brain barrier. The concept of the blood–brain barrier (BBB) was proposed as early as 1900 by Lewandowsky14 (reviewed15) but was not fully appreciated for many decades to come. This means that most polar agents (i.e., those that are not very lipophilic – simple phenols, catechols, carboxylic acids, quaternary amines) typically fail (or display a reduced ability) to gain entry to the brain.

Monoaminergic Activity Enhancer
Taking phenethylamine along with meperidine (Demerol) might cause too much serotonin in the brain and serious side effects including heart problems, shivering, and anxiety. Desipramine (Norpramin) can affect how quickly this protein clears phenethylamine. Taking phenethylamine along with desipramine (Norpramin) might increase the levels and the effects of phenethylamine. This might cause too much serotonin in the brain and could result in serious side effects including heart problems, shivering, and anxiety. Phenethylamine stimulates the body to make certain chemicals that play a role in depression and other psychiatric conditions. People who don’t make enough phenethylamine naturally may be helped by taking phenethylamine as a supplement.
125IDOI, with higher specific activity, and DOI not being a controlled substance, has seen wider application and is currently commercially available and fairly widely used in binding studies. 5-HT2A binding data are provided for several representative agents in Table 1. Over the years, there have been many attempts to develop animal models of hallucinogenic drug action. Described below is a procedure we have found useful for our studies, but it is not an animal model of hallucinogenic drug action; rather, it is a model of stimulus similarity. Non-psychoactive phenethylamines include bronchodilator medications used for asthma such as albuterol while psychoactive phenethylamines include psychostimulants such as amphetamine, methamphetamine, and cathinones (‘bath salts’).
Many neurohackers prolong the effects of PEA by using it with a monoamine oxidase-B inhibitor (MAOI) like selegiline (L-Deprenyl), hordenine, or Oat Straw. The half-life of PEA taken as a nootropic supplement is only 5 – 10 minutes.v Because it’s quickly degraded by monoamine oxidase-B (MAO-B). While it’s unlikely to when taken in moderate doses, in high doses it may cause a positive test result for amphetamine/methamphetamine. It can be a good source of amino acids for plant-based eaters, but it’s not an alternative to taking phenylethylamine supplements. In addition, at least 5 deaths have been reported in people who had taken “legal” phenethylamine-based drugs 64. The US Food & Drug Administration (FDA) has issued warnings to supplement manufacturers that their products contained such forms of phenethylamines called beta-methylphenethylamine (BMPEA).
Before leaving the topic of fundus 5-HT receptors, it is worth mentioning that attempts were made to determine whether peripheral rat fundus 5-HT receptors bore any similarity to brain 5-HT1 or 5-HT2 receptors. The concept of tryptamine receptors had been around for a number of years and it had been speculated that rat fundus might possess both 5-HT and tryptamine receptors. Cohen and Wittenaur25 examined this in detail and concluded that both 5-HT and tryptamine contracted rat fundus by acting on the same population of 5-HT receptors, but via receptors that were distinct from brain 5-HT1 or 5-HT2 receptors. This caused us to seriously question the relationships we had identified between peripheral fundus 5-HT receptor affinity and human hallucinogenic action or central DOM-like stimulus activity.
- We believe that the most accurate information is found directly in the scientific source.
- And, this conclusion was supported by being able to train the same group of animals to discriminate between the two optical isomers.
- PEA binds to C-protein-coupled receptors TAAR1 and TAAR2, receptors reserved specifically for trace amine use.iv These receptors are not used by other major neurotransmitters like dopamine or norepinephrine.
- Patients whose symptoms improved in response to treatment with methylphenidate had a significantly higher PEA level than patients who did not experience such an improvement in their condition (Kusaga et al., 2002).
13 Monoamine Oxidase (MAO) Receptors
Phenylisopropylamines (i.e., α-methyl analogs of phenylethylamines) are a class of phenylalkylamines that includes amphetamine (8) and methamphetamine (52; Figure 11). By introduction of the β-OH function, phenylpropanolamines now possess two chiral centers and four optical isomers are possible (Figure 11). Depending upon stereochemistry, norephedrine is the β-OH analog of amphetamine whereas ephedrine is the β-OH analog of methamphetamine; the other two optical isomers are referred to using the prefix “pseudo” (Figure 11). The naturally-occurring (-)ephedrine (53), the most potent of the phenylpropanolamines as a central stimulant, is several times less potent than amphetamine or methamphetamine. Later, working with Dr. Milt Teitler (Albany Medical College), we found a significant correlation between the stimulus generalization potency of various agents in DOM-trained animals and rat-brain homogenate 5-HT2 receptor affinity (see next section).
Pages In Phenethylamines
Among the various neurotransmitters influenced by PEA, dopamine stands out as particularly significant. Dopamine plays a crucial role in our brain’s reward and pleasure centers, influencing everything from motivation and learning to motor control and emotional responses. The relationship between PEA and dopamine is so intertwined that understanding one inevitably leads to insights about the other. One of the key features that make PEA such a potent neuromodulator is its ability to cross the blood-brain barrier.



